|
Students filled in a general outline for solutions and we discussed specific examples. They also worked to complete a WebQuest on Osmosis & Diffusion. We will go into more detail after Thanksgiving.
0 Comments
Due to a short schedule today we didn't get quite as much covered. We began with Day 3 of the Egg Experiment. They again measured the volume of liquid in the beaker and the mass of the egg. After placing the egg in the beaker again, each student added 100mL of distilled water. On Day 3, students found that that the liquid increased in volume and that the mass of the egg decreased.
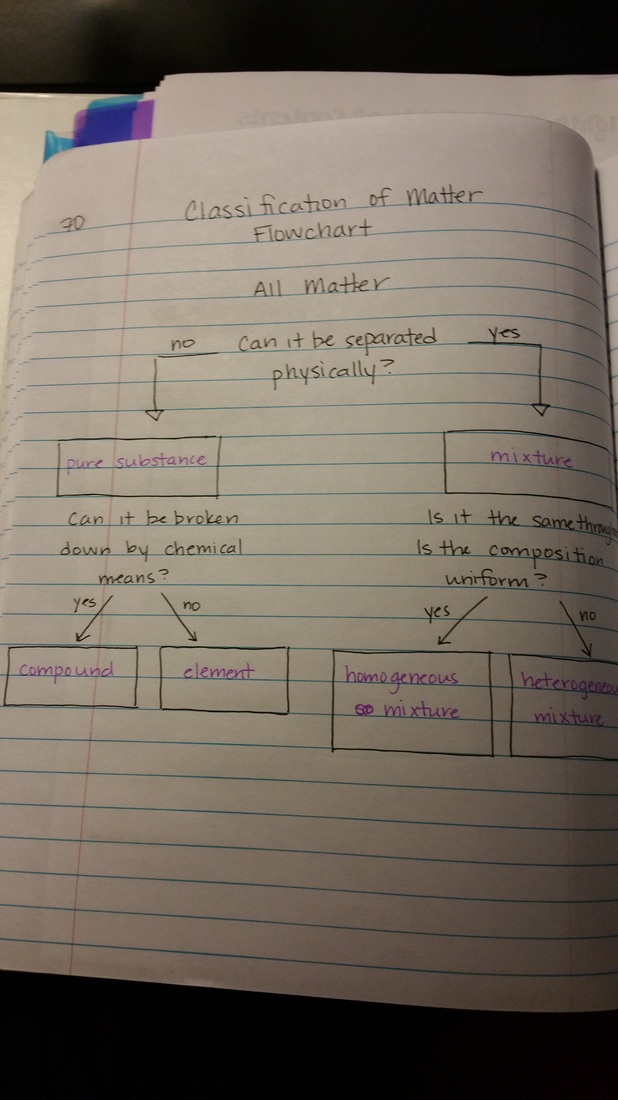
We also added and completed the Matter Flow Chart to give a general overview of the unit and where we are at the moment. Students completed Day 2 in our Egg Experiment. They measured the volume of liquid in their beaker and the mass of the egg. The egg was placed back in to the beaker and we added 75-100mL of corn syrup. Students observed first the the shell seemed to have dissolved away. They also noticed that the liquid around the egg was less than what they put in and that the eggs mass increased.
Today was a busy day in the classroom. Students worked to complete a lab on determining Pure Substances vs. Mixtures. They were given eight different substances to test. They were able to take some of the substance out and place it in a beaker. Students were given water, a loupe, a magnet, black paper, and white paper to perform tests to determine if a substance was pure or a mixture.
Each group began with a conversation about what a pure substance is and based on that definition, how they will test the substances. The groups were reminded that the goal was not to determine what the substance is but to decide if it is pure or a mixture. Students took their test on compounds today as well as began the week long experiment "Can an Egg Bounce?" We began by taking the mass of a raw egg, placing it in a beaker and then adding 100-150mL of vinegar to it.
We went over the review stations from Tuesday and began our new unit today. Students listed the Essential Questions that this unit will answer as well as reviewing the overall task. This unit, students will create a lab report covering solutions, osmosis, diffusion and solubility.
Essential Questions: 1. What is the difference between a pure substance and a mixture? 2. Compare and contrast a heterogeneous and homogenous mixture. 3. What is a solution in terms of solute and solvent? 4. What is diffusion and osmosis? 5. Explain hypertonic, hypotonic, and isotonic. 6. Identify and explain a solution's concentration. Students completed review stations today to further practice and prepare for the Compounds test on Friday. Students finished up Bonding Basics Review and Justified Statements to help prepare for our test on Friday
After a bit more practice on Ionic Bonding we moved on to Covalent bonding. Students learned that these are different than ionic bonds in how their electrons interact. Students also learned a third type of bonding-metallic bonds that form between metals. Students began an overall review of compounds to prepare for their test which will be next Friday, November 13.
Today we continued our study of Ionic bonds by working on drawing more examples and learning how to name them.
To draw an ionic bond we learned: 1. Draw the Lewis Dot Structure for each element. 2. Use arrow(s) to show the loss/gain of electrons 3. Write the ions for each element (remember this should equal zero when added together) 4. Write the chemical formula 5. Write the name |
AuthorScience teacher in small town Alabama. Southern girl raised in SC & GA. I believe that everyone can learn and everyone can find something they like about science. Archives
September 2016
Categories |

















 RSS Feed
RSS Feed
